Dry eye: an originalist perspective
Virtually all patients encounter streams of salty water coursing down their cheeks early in life and don’t realise they have more than one type of tears. More widely recognised, reflex tears serve as a natural emergency eyewash, however, these are very different to the structured basal tears that maintain vision and ocular surface protection.
The tears and our external eye structures are marvels of human engineering that coexist for one simple reason – to provide a smooth and stable refractive surface overlying the relatively irregular corneal epithelium. Our stable tear film and acute vision are among the primary reasons why man has become the dominant species on earth. And such an originalist view is key to understanding both function and dysfunction of the tears and ocular surface.
Why ‘dry eye’?
The term ‘dry eye’ remains in common use despite being a poor description of the disease. A truly dry eye would be opaque and dysfunctional. Thankfully, most ‘dry eyes’ remain reasonably functional. In 2006, the Delphi panel suggested the use of the term tear dysfunction rather than dry eye[1] and Korb and Blackie described dry eye as the wrong diagnosis for millions in 2015[2].
In a practice limited to dry eye in extremely hot and dry Phoenix, Arizona, I rarely, if ever, see purely aqueous deficient dry eye, including patients suffering from Sjögren’s syndrome and other immune disorders. The majority of my patients have meibomian gland dysfunction (MGD) and other contributing factors as major drivers of their misery.
Our continued rigid differentiation of dry eye into aqueous deficient or evaporative subtypes adds to the confusion. On a clinical continuum, what we usually define as aqueous deficient dry eye is often at least partially caused by or worsened by evaporation rather than simply by a lack of aqueous production. While what we describe as evaporative dry eye is more complex than simple evaporation and often a result of tear instability caused by a combination of MGD and enzymatic degradation of tear lipids due to lid bacteria overpopulation, usually associated with MGD. Thus, for the overwhelming majority of dry eye patients, their problem is not a lack of tears, but rather dysfunction of the tears they do have.
Causes of tear dysfunction
MGD is well recognised as both a cause and a contributor to dry eye. It is an obstructive and inflammatory disease that results in insufficient and abnormal production of tear lipids. While most associate MGD with excessive tear evaporation, its role in tear dysfunction is far more complex. Meibomian gland lipids are extremely complex and provide a critical element in tear structure. Hydrophobic bonding of these lipids creates a tarpaulin like covering of the ocular surface. Phospholipids in meibum possess both hydrophobic and hydrophilic properties and help bond outer nonpolar lipids to the underlying hydrophilic tear structure, and all have important tear stabilising functions well beyond being an evaporative barrier. While we understand the composition of tear lipids in general terms, we are years away from assaying and understanding individual variation and its impact on tear structure and function.
An unstable tear structure leads to surface exposure and, in more severe cases, damage. Maintaining the integrity and function of the ocular surface is so critical the eye is equipped with a variety of mechanisms to maintain and regain homeostasis. Inflammation, which plays an important role in the progression of tear dysfunction, results when balance is not achieved and can lead to even greater dysfunction. However, inflammation is a result and, with the exception of autoimmune disease, rarely a cause. Unstable tears also provide a poor refracting surface, another frequent complaint associated with ‘dry eye’.
Obstruction is the most widely recognised cause of MGD and resulting tear lipid insufficiency. As meibum stagnates and becomes increasingly saturated and thickened, pressure within the glands mounts and production is down-regulated. In more severe cases, gland tissue is lost, most likely through inflammation as the body attempts to clear rancid lipids within the glands.
Gland clearance has long been used as a treatment for MGD. Performed appropriately, this includes the application of heat to help melt congealed meibum and mechanical expression. The regular use of warm compresses and massage, expression of the glands with forceps or paddles and especially, the use of advanced technology like automated meibomian gland expression or intense pulsed light (IPL) can be extremely helpful in managing patient signs and symptoms. However, based on my experience, understanding and addressing the underlying causes of MGD, lipid deficiency and tear instability are an important foundation that can improve patient outcomes dramatically.
Overlooked causes and cures
Changes in the visual environment - The ocular surface and tear film were designed to meet the needs of hunter gatherers and didn’t anticipate Guttenberg and his printing press or Steve Jobs and the iPhone. Both book reading and computer use result in decreased blink rates and increases in partial blinks[3],[4]. Since meibum release depends upon normal lid function, including full and complete blinking, decreased clearance and stagnation of meibum is a likely consequence of the modern visual environment with excessive digital device use. Blinking exercises can be beneficial, but the importance of reestablishing normal blink patterns must be reinforced as an essential part of restoring function to the meibomian glands.
Our shifting diet – Think of the meibomian glands as you would a wood stove. Without wood, no heat would be produced. Likewise, the meibomian glands require essential fatty acids (EFAs) as raw materials to produce the complex lipids that make up meibum[5]. Over the past century, significant changes in our intake of EFAs have increased the ratio of omega-6 to omega-3 fatty acids[6]. Despite the controversy of the recent DREAM study[7], general consensus and personal experience reinforce the importance of omega-3 supplementation[8]. After significant in-office testing, I currently recommend 2.5g/daily of a re-esterified triglyceride formulation containing a 3:1 EPA to DHA ratio or greater.
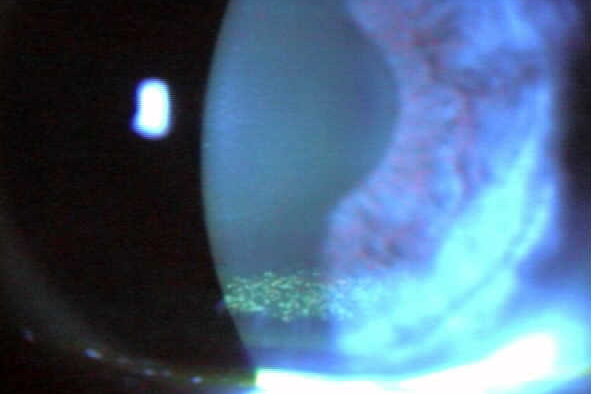
Altered lid microbiome and enzymatic degradation of tear lipids - MGD is associated with alterations in the lid microbiome[9]. Biofilm accumulation and significant bacterial overpopulation of the lids with subsequent release of bacterial toxins and elaboration of lipase and other bacterial digestive enzymes occurs[10]. Staphylococcal toxins target competing species, but they are pro-inflammatory and cause irritation and inflammation when they get into the eye. Bacterial elaborated lipase in the tears breaks down and saponifies meibum producing soaps that cause burning, tear frothing, that can be observed via a slit lamp (Fig 2), and the degradation of tear structure and stability[11]. This often occurs and is easily missed by the clinician who fails to recognise the importance of the altered microbiome and lid staph overpopulation.
Management is straightforward, requiring hypochlorous acid sprayed directly onto the closed lids and surrounding periorbital skin[12]. Hypochlorous acid is naturally occurring, produced by neutrophils to counter infection and inflammation[13]. It also disrupts bacterial digestive enzymes like lipase and has been used successfully to treat necrotising fasciitis as well as blepharitis and MGD[14].
Lagophthalmos and nocturnal exposure – Complete eye closure at night is important not just to create a seal preventing tear evaporation, but also to allow the ocular surface time to regenerate after extended environmental exposure[15]. In my experience nocturnal exposure occurs in as many as 40% of patients presenting with dry eye complaints.
Exposure is easily discovered simply by asking the patient if they experience dryness in the middle of the night or upon waking. It is confirmed by assessing lid closure by observation with a transilluminator or penlight with the head tilted back about 30 degrees. It is more severe in patients with greater tear instability or those using forced air breathing devices that seal poorly and blow air over the exposed ocular surface.
Management for poor nocturnal lid closure has traditionally been petrolatum-based ointments, however, they are only variably effective, extremely messy and hard to remove. More effective are silicone night masks which create a tight seal and maintain high moisture levels throughout the night. Restoring effective protection at night can produce dramatic improvements with substantial reduction of symptoms on waking and throughout the remainder of the day.
In conclusion
What we call dry eye appears simple but is extremely complex reflecting the critical role the tears and ocular surface play in human function and survival. An overly simplistic view often results in clinical failure, but an over-complicated approach is equally perilous.
Understanding the underlying disease and targeting the causes of tear dysfunction rather than the subsequent signs and symptoms is a much more effective strategy for managing this rapidly growing patient population, and the potential benefits for both patient and practitioner are substantial.
References
-
Behrens A, Doyle JJ, Stern L, et. al. Dysfunctional tear syndrome: a Delphi approach to treatment recommendations. Cornea. 2006 Sep;25(8):900-7. ↑
-
Korb DR, Blackie CA. "Dry Eye" Is the Wrong Diagnosis for Millions. Optom Vis Sci. 2015;92(9):e350-4. ↑
-
Jaiswal S, Asper L, Long J, et. al. Ocular and visual discomfort associated with smartphones, tablets and computers: what we do and do not know. Clin Exp Optom. 2019 Jan 21. ↑
-
Sheppard AL, Wolffsohn JS. Digital eye strain: prevalence, measurement and amelioration. BMJ Open Ophthalmol. 2018; 3(1). ↑
-
Liu Y, Kam WR, Sullivan DA. Influence of omega 3 and 6 fatty acids on human meibomian gland epithelial cells. Cornea. 2016; 35(8): 1122–1126. ↑
-
Blasbalg TL, Hibbeln JR, Ramsden CE, et. al. Changes in consumption of omega-3 and omega-6 fatty acids in the United States during the 20th century Am J Clin Nutr. 2011; 93(5): 950–962. ↑
-
The Dry Eye Assessment and Management Study Research Group. n–3 Fatty Acid Supplementation for the Treatment of Dry Eye Disease. N Engl J Med. 2018 May 3; 378(18): 1681–1690. ↑
-
Epitropoulos AT, Donnenfeld ED, Shah ZA, et. al. Effect of Oral Re-esterified Omega-3 Nutritional Supplementation on Dry Eyes. Cornea. 2016; 35(9): 1185–1191. ↑
-
Suzuki T. Inflamed Obstructive Meibomian Gland Dysfunction Causes Ocular Surface Inflammation. Investigative Ophthalmology & Visual Science November 2018, Vol.59, DES94-DES101. ↑
-
Rynerson JM, Perry HD. DEBS – a unification theory for dry eye and blepharitisClin Ophthalmol. 2016; 10: 2455–2467. ↑
-
Dougherty JM, McCulley JP. Bacterial lipases and chronic blepharitis. Invest Ophthalmol Vis Sci. 1986; 27: 486–491. ↑
-
Stroman DW, Mintun K, Epstein AB, et. al. Reduction in bacterial load using hypochlorous acid hygiene solution on ocular skin. Clin Ophthalmol. 2017; 11: 707–714. ↑
-
Kevin M. Rigby KM, DeLeo FR. Neutrophils in innate host defense against Staphylococcus aureus infections. Semin Immunopathol. 2012; 34(2): 237–259. ↑
-
Epstein A, Pang L, Najafi-Tagol K, et. al.; Comparison of Bacterial Lipase Activity in the Presence of Eye Lid Cleansers. Invest. Ophthalmol. Vis. Sci. 2015;56(7 ):4446. ↑
-
Katz J, Kaufman HE. Corneal exposure during sleep (nocturnal lagophthalmos). Arch Ophthalmol. 1977;95(3):449-53.Optometrist and ocular disease researcher, Dr Art Epstein is a native New Yorker who now resides in arid Phoenix, Arizona where he co-founded Phoenix Eye Care. He heads up the practice’s Dry Eye - Ocular Surface Disease Centre and is clinical research director of Ophthalmic Research Consultants of Arizona. A prolific author, he has published many hundreds of articles, scientific papers and book chapters and is a sought after international speaker on all things dry eye. ↑