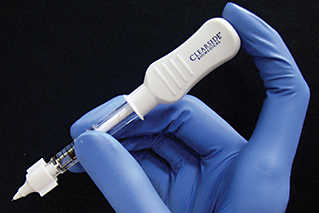
Xipere green-lit by FDA
Xipere, the first product for injection into the suprachoroidal space to treat macular oedema associated with uveitis, has been granted US Food and Drug Administration (FDA) approval.
Developed by Bausch + Lomb, Xipere (triamcinolone acetonide) is delivered to the back of the eye using Clearside Biomedical’s SCS Microinjector to provide compartmentalised delivery and better absorption, according to a joint statement by the two companies.
FDA approval followed Xipere’s phase-3 clinical trial of 160 patients, in whom best-corrected visual acuity (BCVA) had improved by at least 15 letters from baseline and a statistically significantly greater proportion of treated patients (47%) achieved at least a 15-letter improvement in BCVA than patients in the control arm (16%, p< 0.01) after 24 weeks of follow-up, according to the companies.