Contact lens-related keratitis in New Zealand
In the early Sixties, driven by the efforts of pioneering dentist-turned-contact lens manufacturer Eugene Hirst (original co-founder of Mortimer Hirst in Auckland), New Zealand was the first country outside Czechoslovakia to work with the first viable, hydrophilic soft contact lens (CL) material hydroxyethyl methacrylate (HEMA)*. Though a breakthrough for many people unable to wear glasses, research in Canada soon showed that bacteria could grossly contaminate the polymer matrix of soft contact lenses1.
Since then, contact lens-related microbial keratitis (CLMK) has blighted corneas at an established incidence worldwide of 2-4 per 10,000 per year for daily soft CL wearers, rising 10-20 times for overnight wearers2. Despite advances in material biocompatibility, rates of infection have not reduced2.
Correlating microbiology, pathophysiology and clinical features
CLMK is considered a subset of corneal infiltrative events where active microbial replication is implicated in the pathogenesis, differentiating it from other microbial-mediated processes such as bacterial exotoxin-induced hypersensitivity (staphylococcal hypersensitivity). CL-related bacterial keratitis establishes through an epithelial breach (such as the microtrauma of CL wear). The resultant acute infiltrative event is a cellular aggregate of polymorphs typically appearing within four to six hours of symptom onset. The initial infiltrate is primarily subepithelial and indistinct, with limbal haze from cellular migration, eventually coalescing to form a focal opacity with an overlying epithelial defect. Depending on severity, surrounding inflammation can be seen as bulbar injection/chemosis, anterior uveitis and, ultimately, lid oedema. Progression of the infection leads to ulceration, and if particularly aggressive, corneal perforation.
In contrast, non-infectious hypersensitivity reactions tend to produce minimal or no epithelial defect relative to the underlying infiltration, since the defects emerge from inflammatory breakdown rather than aggressive microbial invasive mechanisms. As there is considerable overlap in non-infectious and infectious presentations, accurate documentation of the dimensions of the infiltrate, epithelial defect, location, maximal stromal thinning and anterior chamber reaction is vital for determining severity and assessing clinical response.
Care should be taken to ensure the slit lamp beam is perpendicularly aligned and in focus to ensure the measurements are not falsely increased from projection errors. Corneal sensation should be checked in all cases of keratitis, although this is usually slightly reduced in long-term CL wearers and may be of limited positive predictive clinical value unless greatly diminished.
Over 90% of CLMK are caused by bacteria2 with the gram negative, water-borne Pseudomonas aeruginosa being the most prevalent in most regions3 and capable of perforating the cornea within days. P. aeruginosa is well adapted for CLMK, capable of adhering and replicating in vitro in soft lens materials, forming biofilms which confer resistance to environmental challenges, and establishing infection through invasive cellular or cytotoxic mechanisms2.
Acanthamoeba keratitis, almost exclusively seen in CL wearers, carries a much higher visual morbidity than bacterial CLMK and thus warrants special mention. The protozoan Acanthamoeba is ubiquitous in the environment, distributed in air, soil and water sources. The ability to transform into chemically resistant cysts in response to challenging environments allows Acanthamoeba to survive in chlorinated swimming pools and most CL multipurpose cleaning solutions4. Local data in Auckland show rising cases of Acanthamoeba keratitis peaking in the summer months from February to April5. Whether this suggests a climactic response in virulence or a higher rate of casual CL wear with high-risk behaviours has yet to be elucidated. Diagnostic delay is common, as the classic ring infiltrate with radial perineural infiltrates is only present in the late stages, whilst the early presentation of punctate epithelial erosion is commonly misdiagnosed as contact lens overwear (Fig 1). Other features overlapping with bacterial CLMK include stromal infiltrate (40%), anterior chamber reaction (29%) and epithelial defect (76%)5. Therefore, a high index of suspicion is required in all CLMK and begins with documentation of Acanthamoeba specific risk factors including hot-tub use, swimming and use of tank water supply5. Empirical therapy with antibiotics for bacterial CLMK usually leads to an initial improvement of symptoms (depleting the cornea of Acanthamoeba’s main nutritive source, bacteria), followed by a relapse once treatment is ceased.
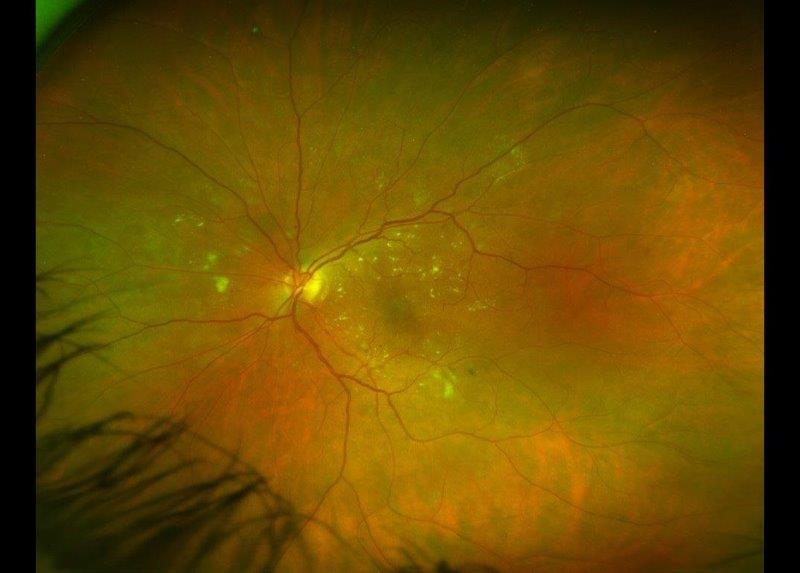
The majority of fungal CLMK infections are caused by Fusarium, Aspergillus and Candida species2, with an insidious course similar to Acanthamoeba keratitis. Fungal species exhibit a diverse range of morphologies and a constellation of presentations can be seen, including the classic description of a poorly defined fluffy infiltrate edged with satellite lesions (Fig 2). Extensive and deeply buried hyphae pose special challenges as they are too large to be ingested by host immune cells and require deeply penetrating antifungal formulations. The main risk factor is ocular trauma involving organic matter6 and thus a history of trauma should be documented. Thankfully, New Zealand has relatively low rates of fungal keratitis, with 2-5% positive on corneal scrapes3, 7.
Fig 2. Fusarium keratitis with ill-defined infiltrate, satellite lesions and hypopyon
Rationalised community treatment and follow up
There is little doubt of the diagnosis of bacterial CLMK in the contact lens wearer that presents with acute ocular pain, redness, photophobia, purulent discharge, a large infiltrate with intense staining and hypopyon. However, CLMK frequently presents with less remarkable signs and symptoms and is often difficult to differentiate from a sterile infiltrative event (Fig 3). Because of the potential poor or severe outcome of missing a bacterial CLMK, the preferred management of these cases is to start empirical antimicrobial treatment.
Fig 3. Sterile or infective CL keratitis?
Contemporary microbiological data guides first-line therapy. All positive corneal scrapes (n=89) at Auckland’s Greenlane Clinical Centre from 2013-2014 were reviewed by Marasini et al indicating Pseudomonas, Staphylococci, Streptococci, Moraxella and Corynebacterium species account for 82.5% of all cultured species8. Ciprofloxacin (a first-generation quinolone) remains an appropriate choice for topical monotherapy with 100% gram-negative and 98.8% gram-positive coverage8. Dual therapy with topical cephalosporin and topical aminoglycoside is an alternative treatment regimen typically reserved for severe keratitis, although similar outcomes have been demonstrated comparing dual vs mono therapy9. Auckland’s dual-therapy protocol for severe keratitis requiring admission utilises non-preserved compounded cefuroxime 5% (second-generation cephalosporin) and tobramycin 1.36% hourly. Marasini’s data has shown that cefazolin (first-generation cephalosporin), ceftazidime (third-generation cephalosporin) and gentamycin and tobramycin (aminoglycosides) provide 100% coverage of the bacterial species cultured8. Cefuroxime shows poor activity against gram-negative species (only 33% being susceptible)8. It should be noted, however, that local published data on positive scrapes are often limited to severe or atypical keratitis and there has been no sub analysis for CLMK.
In the setting of a small (<1mm) peripheral infiltrate with no significant anterior chamber reaction, the frontline clinician can judiciously prescribe outpatient first-line empirical treatment with intensive (hourly) ciprofloxacin 0.3% during daytime hours. Chloramphenicol 1% ointment at bedtime is commonly prescribed alongside ciprofloxacin, however there is no evidence of improving outcomes with this practice and it lacks activity against Pseudomonas10. In this scenario the patient should be reviewed every 24-48 hours and if improvement in symptoms and inflammation is noted after 36-48 hours of intensive treatment, then the frequency of ciprofloxacin can be reduced to two-hourly and titrated according to response from there. Excess ciprofloxacin may precipitate into an ulcer (Fig 4) and impede re-epithelialisation. If there is no improvement/worsening of symptoms or inflammation after 36-48 hours, then the patient should be referred for further investigation and intensive treatment with dual therapy. Re-epithelisation of the cornea is a significant sign of improvement and response to treatment, but resolution of the infiltrate can take three weeks or more11 and it can be difficult to clinically differentiate between active infiltrate and organising scar.
Fig 4. Ciprofloxacin precipitates in keratitis seen as white deposits
Once there is reduced infiltrate opacity with full resolution of symptoms, inflammation and epithelial defect, at least another two weeks of contact lens cessation is recommended. Returning to CL wear should be done under the supervision of an optometrist with a refit appointment. Education of modifiable risk factors such as showering, tap water use, overnight wear, CL reuse, smoking, online ordering of CLs and poor hygiene should be undertaken prior to considering a refit. Some patients may no longer be suitable for CL wear in the setting of repeat CLMK or vision threatening sequelae.
Red flags for referral
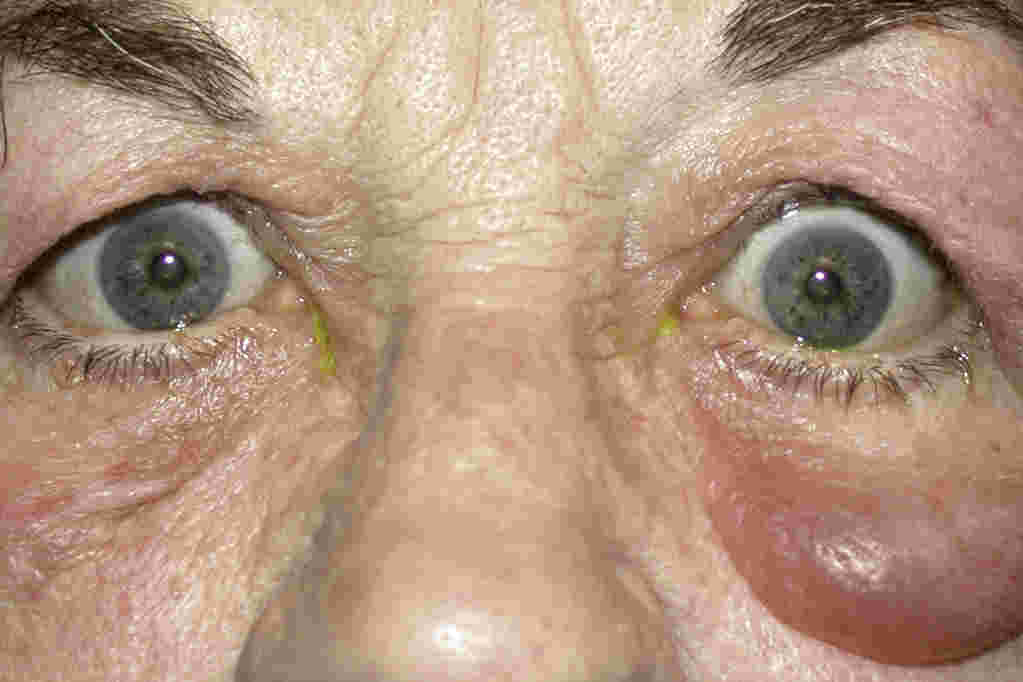
Severe or high-risk CLMK, characterised by a central ulcer, infiltrate >2mm, hypopyon (Fig 5), non-response after 48hrs of intensive therapy, or risk of Acanthamoeba/fungal keratitis, should be referred to a tertiary centre for specialist management. CLMK cases are accepted for review on the same day as referral, since treatment delay over 12 hours carries an increased likelihood of visual loss, duration of infection and cost of treatment12.
A key component of managing microbial keratitis is microbial identification, so corneal scraping is a crucial diagnostic tool. The infiltrate is debrided with a #15 surgical blade or sterile needles and inoculated onto gram-stain glass plates, agars and broths, and a full panel of PCR swabs are taken to assess for bacteria, Acanthamoeba, fungi and viral sources. False positives can occasionally occur from culturing normal ocular flora in up to 10% of cases13 and false negatives can occur with inadequate scraping, use of preserved topical anaesthetic and antibiotic use within the preceding 24 hours. Acanthamoeba culture takes several days and has low scrape sensitivity (22.4%), therefore in-vivo confocal microscopy is a useful adjunct to identify Acanthamoeba cysts5, 14.
Referred cases are assessed and admitted to the ward, if required, for intensive day and night treatment with topical therapy (usually with dual therapy, as described above), as well as adjunctive treatments such as cyclopentolate and oral doxycycline for the associated anterior chamber reaction and to reduce corneal thinning.
Fig 5. Severe contact lens related keratitis with hypopyon and large infiltrate involving the central cornea
References
- Cornea and Contact Lens Society. History of the New Zealand Contact Lens Society 1958-2007[accessed 2 Mar 2021] Available from: https://www.contactlens.org.nz/history
- CarntN,Samarawickrama C, White A, Stapleton F. The diagnosis and management of contact lens-related microbial keratitis. Clin Exp Optom. 2017;100(5):482-93.
- Wong T,Ormonde S, Gamble G, McGhee CN. Severe infective keratitis leading to hospital admission in New Zealand. Br J Ophthalmol. 2003;87(9):1103-8.
- deLacerda AG, Lira M. Acanthamoeba keratitis: a review of biology, pathophysiology and epidemiology. Ophthalmic Physiol Opt. 2021;41(1):116-35.
- McKelvieJ, Alshiakhi M, Ziaei M, Patel DV, McGhee CN. The rising tide of Acanthamoeba keratitis in Auckland, New Zealand: a seven-year review of presentation, diagnosis and outcomes (2009-2016). Clin Exp Ophthalmol. 2018;46(6):600-7.
- Thomas PA,Kaliamurthy J. Mycotic keratitis: epidemiology, diagnosis and management. Clin Microbiol Infect. 2013;19(3):210-20.
- Pandita A, Murphy C. Microbial keratitis in Waikato, New Zealand.Clin Exp Ophthalmol. 2011;39(5):393-7.
- MarasiniS, Wang MT, Swift S, Dean SJ, Ormonde SE, Gamble GD, et al. Clinical and microbiological profile of Pseudomonas aeruginosa keratitis admitted to a New Zealand tertiary centre. Clin Exp Ophthalmol. 2018;46(4):441-4.
- McDonald EM, Ram FS, Patel DV, McGhee CN. Topical antibiotics for the management of bacterial keratitis: an evidence-based review of high quality randomised controlled trials.Br J Ophthalmol. 2014;98(11):1470-7.
- Feder HM, Jr., Osier C,Maderazo EG. Chloramphenicol: A review of its use in clinical practice. Rev Infect Dis. 1981;3(3):479-91.
- Sweeney DF,Jalbert I, Covey M, Sankaridurg PR, Vajdic C, Holden BA, et al. Clinical characterisation of corneal infiltrative events observed with soft contact lens wear. Cornea 2003;22(5):435-42.
- KeayL, Edwards K, Naduvilath T, Forde K, Stapleton F. Factors affecting the morbidity of contact lens-related microbial keratitis: a population study. Invest Ophthalmol Vis Sci. 2006;47(10):4302-8.
- Sharma S,Kunimoto DY, Gopinathan U, Athmanathan S, Garg P, Rao GN. Evaluation of corneal scraping smear examination methods in the diagnosis of bacterial and fungal keratitis: a survey of eight years of laboratory experience. Cornea 2002;21(7):643-7.
- Patel DV, Zhang J, McGhee CN. In vivo confocal microscopy of the inflamed anterior segment: A review of clinical and research applications.Clin Exp Ophthalmol. 2019;47(3):334-45.
* A historical look at the first ‘soft’ contact lenses being made by Hirst Contact Lens Laboratory, featuring Eugene Hirst, circa 1949. Credit: Mortimer Hirst, MOTAT - https://www.youtube.com/watch?v=X0r77oJBrng
Dr William Shew is a recent non-vocational registrar at the acute eye service at the Auckland District Health Board (ADHB) and a therapeutic optometrist. He is the current neuro-ophthalmology clinical fellow at the University of Auckland.
Dr Sue Ormonde is a senior lecturer at the University of Auckland with extensive experience as a cornea and anterior segment specialist at Greenlane Hospital (ADHB) and in private practice at Auckland Eye.