Luxturna rival emerges
Interim results from the phase 2 trial of Beacon Therapeutics AGTC-501 (laruparetigene zovaparvovec), a gene therapy for patients with retinitis pigmentosa (RP), show it presents a “formidable challenge” to Spark Therapeutics’ Luxturna, according to data analysis firm GlobalData.
Beacon reported 12-month data from its Skyline trial showed AGTC-501 was well-tolerated, with the majority of treatment-related adverse events being mild to moderate in severity and no clinically significant safety events. Vista, the phase 2/3 trial of AGTC-501, is anticipated to begin in the first half of 2024, said the company.
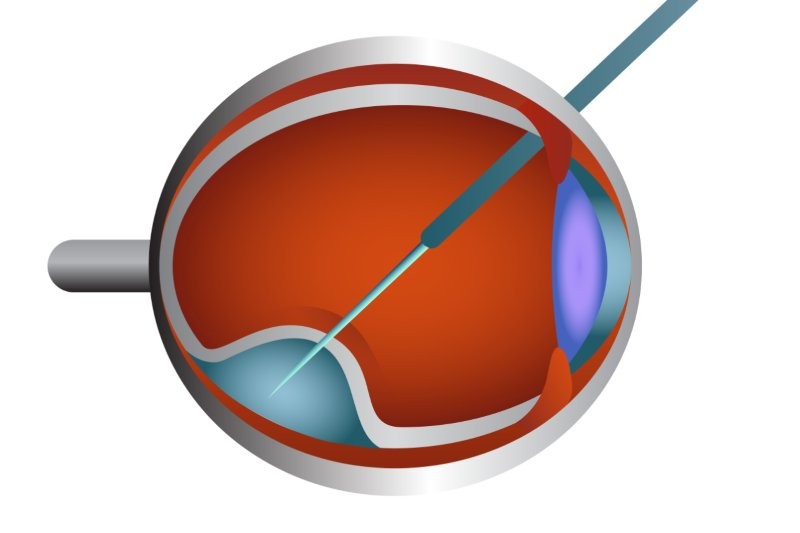
Luxturna (voretigene neparvovec-rzyl) is indicated for the treatment of RPE65-mutation-associated Leber congenital amaurosis (LCA), which is considered more severe than other inherited retinal diseases such as RP, according to Cideciyan and Jacobson (2019). Although approved by Medsafe for use in New Zealand, Luxturna costs NZ$675,000 per eye, excluding surgical expenses. Its current Pharmac funding application has stalled.
AGTC-501 offers an alternative mechanism of action to Luxturna, said Sara Reci, senior pharma analyst at GlobalData. “AGTC-501 is an X-linked retinitis pigmentosa GTPase regulator (RPGR) activator. This has positive implications for patients, which include potentially introducing a second gene therapy into the retinitis space.” AGTC-501 shares its mechanism of action with MeiraGTx’s botaretigene sparoparvovec, which is currently in phase 3 of development, said Reci.