The iris in health and disease: part 1 - normal variations and mass lesions
The healthy human iris consists of five layers: the anterior-border layer, stroma, iris sphincter muscle, iris dilator muscle and the iris pigment epithelium (IPE)1. The anterior border layer is made up of fibroblasts and contains melanocytes1. Underneath that is the iris stroma, a layer that contains a loose mesh of collagen fibres, melanocytes, fibroblasts and clump cells1. This is followed by two muscular layers: 1) the iris sphincter muscle, which surrounds the pupil in a concentric manner and is responsible for pupil constriction, and 2) the iris dilator muscle (consisting of fibres that are arranged radially around the pupil), which is responsible for the dilation of the pupil2. The most posterior layer, the IPE, is dense with melanin-rich melanocytes. The iris is responsible for modulating light and accommodation. Any compromise to the iris can result in symptoms including photophobia, glare, reading difficulty and altered contrast sensitivity. Iris appearances also have a cosmetic effect, varying from one individual to another and can provide evidence to underlying systemic diseases2.
Healthy variations in the human iris include Fuchs’ crypts, pigment spots, Wölfflin nodules and contraction furrows1. Fuchs’ crypts are diamond-shaped, small or large lacunae in the anterior-border layer of the iris and arise from the reabsorption of the pupillary membrane (Fig 1A)1. Pigment spots are small, variable-shaped areas of increased pigmentation occurring anywhere on the anterior border layer and are typically superficial, referred to as freckles. When they distort the underlying stroma, these pigment spots are referred to as naevi and they typically carry an indolent course (Fig 1B) 3. Wölfflin nodules are small, yellow collagen bundles that accumulate at the periphery of the iris occurring in approximately a quarter of the European population (Fig 1C) 1. Finally, contraction furrows are iris folds that resemble wrinkles in the iris peripheries and are thought to be the product of the contraction and dilation of the pupil (Fig 1C) 1.
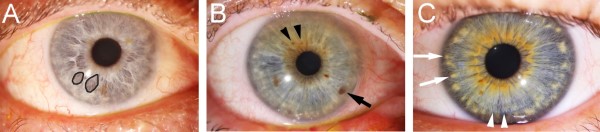
Fig. 1 Healthy variations in the human iris. (A) Fuchs’ crypts (outlined), (B) pigment spots/freckles (black arrowheads) and a naevus (black arrow), (C) Wölfflin nodules (white arrows), and contraction furrows (white arrowheads)
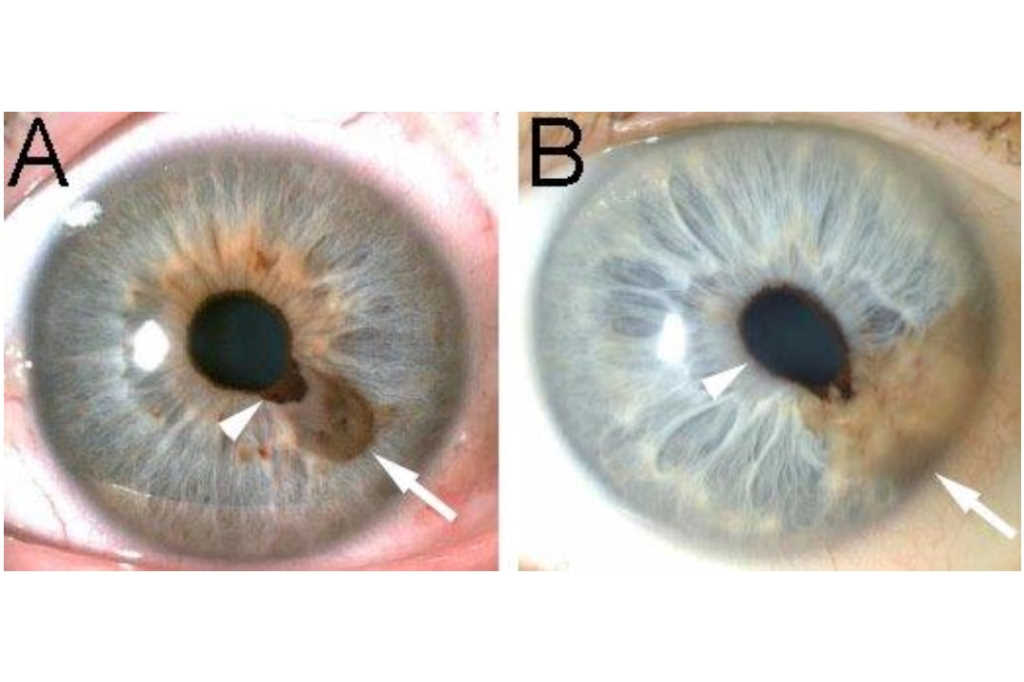
Certain iris appearances are associated with genetic abnormalities, systemic disease or represent a localised malignant transformation. A clinician may find Brushfield spots – small, white or greyish/brown spots on the iris peripheries (Fig 2A) – in individuals with trisomy 212 and these appear clinically similar to Wölfflin nodules. Lisch nodules – clumps of pigment that are 1-2mm in size and appear as yellowish-brown dome-shaped lesions in all zones of the iris (Fig 2B) – can be seen in individuals with neurofibromatosis type 12, an autosomal dominant genetic condition characterised by café-au-lait spots, multiple neurofibromas and skin freckling under the armpits and in skin folds. Individuals with Waardenburg syndrome, another autosomal dominant genetic condition, can present with hearing loss and pigmentary changes in the melanin-producing sites of the body (hair, skin and eye). They may demonstrate unilateral or bilateral complete, partial or segmental heterochromia (Fig 2C)3. A coloboma – a missing segment of the iris resulting in pupil distortion – may indicate an underlying developmental anomaly or chromosomal abnormality such as that found in ‘cat-eye’ syndrome. The presence of a coloboma should prompt further investigation, including genetic analysis (chromosome karyotype) and a complete ocular exam, as the coloboma may involve the retina (Fig 2D).
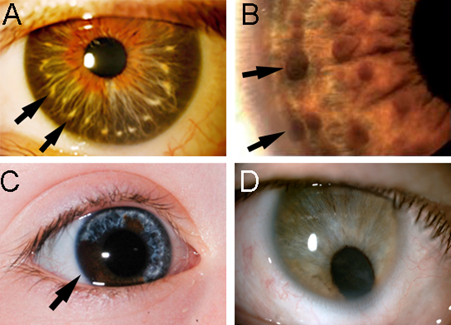
Fig. 2. (A) Brushfield spots (arrows) associated with trisomy 21, (B) Lisch nodules (arrows) associated with neurofibromatosis Type 1, (C) heterochromia associated with Waardenburg syndrome, and (D) congenital iris coloboma with inferior iris defect
Iris nodules when seen together with uveitis can also provide clinicians with clues to underlying systemic disease, including sarcoidosis, multiple sclerosis, metastatic malignancies, lymphoma and leukaemia5. Koeppe (small white/tan nodules seen in the inner margin of the iris) or Busacca (larger white/tan nodules located on the mid periphery region of the iris anterior border layer) – seen in Figs 3A and B – can be found in approximately 10% of patients with sarcoidosis5. Infectious aetiologies such as tuberculosis, bacteraemia or candidaemia, tend to present as large, fluffy or creamy looking iris nodules, alongside other systemic symptoms such as fevers and rigors5.
Granulomatous anterior and/or intermediate uveitis with iris nodules, typically appearing as white nodules on the iris surface or angle (Fig 3C), in an individual with neurological symptoms will need to be worked up for sarcoidosis and multiple sclerosis5. Metastatic malignancies (most commonly from the breast, lung and skin6), lymphoma and leukaemia may present as greyish-white, translucent iris nodules and can be associated with hypopyon, hyphaema, secondary glaucoma and iris atrophy5. These lesions may resemble iris cysts and therefore appropriate history, examination and further investigations are warranted at first presentation4.
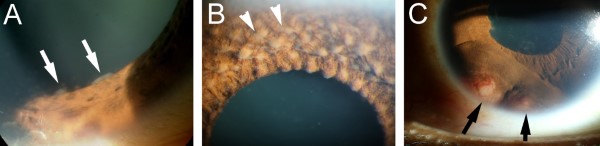
Fig 3. Iris changes associated with systemic diseases. (A) Koeppe nodules found on the pupillary margin (arrows), (B) Bussaca nodules (arrowheads) on the iris anterior border layer, typically in the iris mid-periphery, and (C) iris granulomas (arrows) located close to the angle, associated with sarcoidosis
Lastly, the most common primary malignancy arising from the iris is melanoma, which typically occurs in individuals >40 years old3. Iris melanomas are rare (2-5% of uveal melanomas), typically found in the inferior iris, often pigmented (although a small number are amelanotic) and may be either circumscribed or diffuse (Fig 4)3. Despite the ominous connotation, the overall prognosis is generally favourable, with an exception for diffuse iris melanomas, which are typically diagnosed late and associated with higher risk of metastases3. Diffuse iris melanomas can present with unilateral hyperchromatic heterochromia and glaucoma (the latter secondary to invasion of the iridocorneal angle), which often result in a significant delay in diagnosis3. Iris melanoma can occasionally arise from a benign naevus with a similar appearance, thus referral for a second opinion from a specialist is recommended.3
It is truly remarkable what we can tell about an individual’s health just by looking at the iris.
In 2022, Dr Joevy Lim and colleagues from the University of Auckland, led by Professor Charles McGhee, will conduct a nationwide prospective study on ocular melanocytic lesions to identify characteristics that will help differentiate malignant from benign lesions. We invite all New Zealand optometrists to fill out the following survey so we can provide more details of the study: www.surveymonkey.com/r/F37W83R
The second part of this two-part series explores retro illumination iris defects and conditions causing rubeosis (neovascularisation of the iris). We would like to acknowledge Professor Charles McGhee, Dr Rachael Niederer and Joyce Mathan for their generous contributions of illustrative case images to these articles.
References
- Edwards M et al. Analysis of iris surface features in populations of diverse ancestry. R Soc Open Sci. 2016;3(1):150424.
- Morrison PJ. The iris – a window into the genetics of common and rare eye diseases. Ulster Med J. 2010;79(1):3-5.
- Rennie IG. Don’t it make my blue eyes brown: heterochromia and other abnormalities of the iris. Eye (Lond). 2012;26(1):29-50.
- Shields CL et al. Review of cystic and solid tumours of the iris. Oman J Ophthalmol. 2013;6(3):159-164.
- Myers TD et al. Iris nodules associated with infectious uveitis. Br J Ophthalmol. 2002;86(9):969-974.
- Shields CL et al. Clinical survey of 3680 iris tumours based on patient age at presentation. Ophthalmology. 2012;119(2):407-414.



Dr Joevy Lim is an HRC clinical research fellow and PhD student in the Dept of Ophthalmology, University of Auckland (UoA). Her research focuses on ocular melanoma and melanocytic lesions, and anterior segment disorders.
Dr Jie Zhang is a senior HRC research fellow and scientist in the Dept of Ophthalmology, UOA. She has specific research interests in both laboratory and clinical aspects of the cornea and anterior segment.
Dr Rachael Niederer is a senior lecturer in the Dept of Ophthalmology, UOA and a consultant ophthalmologist with a special interest in clinical and research aspects of uveitic eye conditions.