NZ researchers lead worlds first trial of TED cure
New Zealand researchers are set to conduct ‘Reclaim’, the world’s first trial of Kriya-586, a one-time treatment with the potential to “turn off” thyroid eye disease (TED).
US company Kriya Therapeutics describes its product as an adeno-associated virus gene therapy that drives durable expression of a monoclonal antibody that blocks insulin-like growth factor 1 receptors (IGF-1R). “This decreases pro-inflammatory thyrotropin receptor autoantibody signalling, which may lead to reductions in proptosis and diplopia. IGF-1R blockade has been established as a clinically validated mechanism of action [in patients with TED],” the company said.
That clinical validation comes courtesy of teprotumumab (Tepezza), an FDA-approved treatment for active, moderate-to-severe TED. Administered via intravenous infusion every three weeks for a total of eight doses, it reduces proptosis and inflammation by targeting orbital fibroblasts, according to the European Medicines Agency.
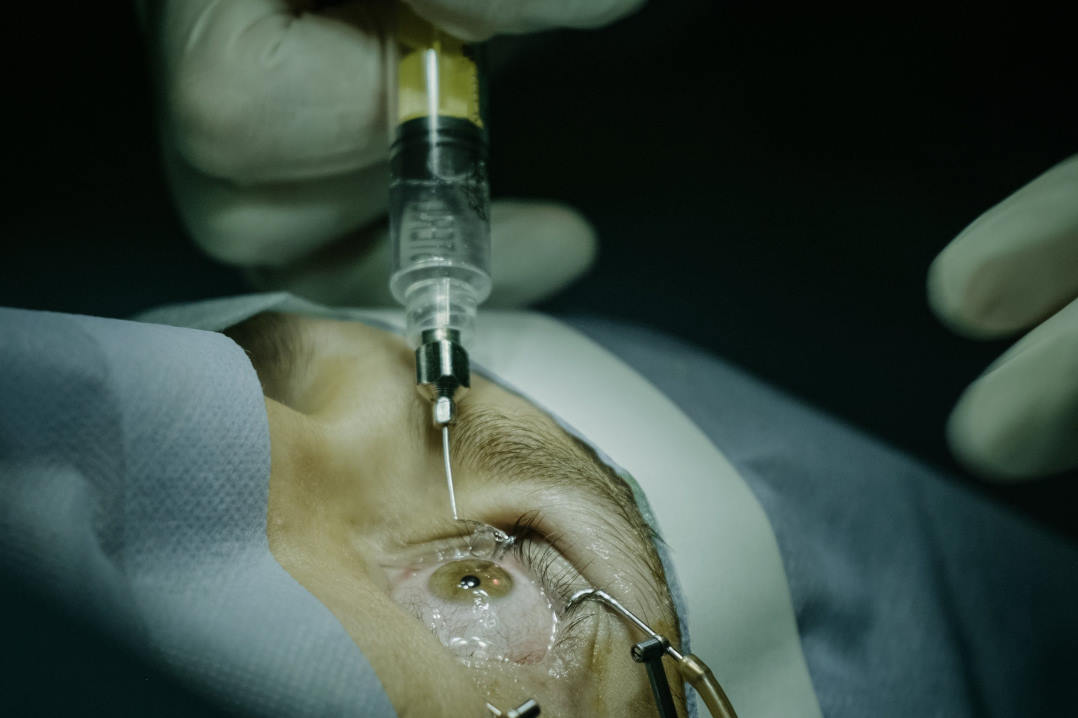
Kriya Therapeutics recognised that local production of teprotumumab might be effective in controlling TED, explained Christchurch’s Dr Rebecca Stack, one of the Reclaim study co-investigators. “[It used the theory that] delivering genetic material to the affected tissue by peribulbar injection will lead to local production of teprotumumab for the life of the orbital fibroblasts… a one-off injection, turning off the disease and curing the patient.”
While teprotumumab has been available in Australia since June 2025, it is not yet available in New Zealand. However, a July 2025 meeting of Medsafe’s Medicines Classification Committee recommended that it be added to the New Zealand Medicines Schedule as a prescription medicine.
“Although teprotumumab has been shown to be effective, studies now seem to show the TED is likely to relapse and likely require re-treatment,” said Dr Stack. It’s also expensive. “The current cost of teprotumumab for a full single course is around US$400,000! I would think it highly unlikely to be [Pharmac] funded.”
“Because New Zealand patients don't have access to teprotumumab, we’ve become a region that appeals to try Kriya-586,” said Dr Stack.
Auckland’s Dr Richard Hart and Wellington’s Dr Jesse Gale have also signed up as co-investigators in the Reclaim study. “Nobody has been recruited yet, but ethics approval has been granted,” said Dr Stack. “We’re just waiting on confirmation of some protocol queries related to where and how the initial injection is to be given. But the site visits, including the Auckland, Wellington and Christchurch centres, have been done.”
According to The New Zealand Medical Journal, New Zealand has a TED incidence rate of up to 11.1 per 100,000 person-years and the risk is double in Māori compared with Europeans. However, even if the New Zealand trial is successful, Kriya-586’s potential use remains restricted. “While the incidence is high, this treatment is aimed at only the severe end of thyroid eye disease – for those with proptosis and/or severe extraocular muscle disease with diplopia. The incidence of moderate to severe thyroid eye disease is only 15% of those affected. So 85% of TED is mild disease, for which Kriya-586 would not be indicated,” said Dr Stack.
At the time of writing, there were no commercially available Kriya Therapeutics products in New Zealand. However, in late 2025, New Zealand Clinical Research began a separate phase 1/2 trial of another product, Kriya-825, a gene therapy for advanced age-related macular degeneration. The therapy is delivered to the suprachoroidal space using the Everads injector, designed to enhance the delivery of gene therapies to the eyes while minimising inflammation, according to its Israeli developer, Everads Therapy.
Dr Stack is encouraging New Zealand eyecare professionals treating patients with active TED to get in touch with her at info@southerneye.co.nz.